LA VIRUELA DEL MONO, OTRA PANDEMIA ?
THE MOKEYPOX, ANOTHER PANDEMIC ?
MONKEYPOX, also known as SIMIAN SMALLPOX (mpox), is a viral ZOONOSIS, transmitted from animals to humans, caused by an ORTHPOXVIRUS, (Monkeypox Virus) related to human smallpox, but with lower transmissibility and lower lethality in humans.
The Virus of HUMAN SMALLPOX, eradicated in the 1980s from planet Earth, also belongs to the ORTHPOXVIRUSES, and is known by the name of Variola Virus (smallpox virus), much more DEADLY (32% or more in unvaccinated), than Monkeypox Virus (8–13% or less). (1)
HISTORY:
The disease was identified for the first time in 1958 during a non‑fatal outbreak in a colony of laboratory macaques in Copenhagen (Denmark), at the Statens Serum Institut (State Serum Institute), by a medical group led by the Danish scientist Preben Christian Alexander von Magnus, a physician born in Denmark (1).
The macaques in question had been imported from Singapore, and they were CRAB‑EATING MACAQUES (Macaca fascicularis, Cynomolgus monkey), used as laboratory animals (1).
In 1958 the scientist and his group observed two outbreaks of smallpox‑like, disease in these colonies of monkeys, which presented cutaneous pustules similar to human smallpox.
The virus was isolated from the kidney of one of these affected monkeys, and from the chorion‑allantoic membrane of chicken embryos, both examined by electron microscope, and it was concluded that it was a new orthopoxvirus, distinct from that of SMALLPOX (smallpox), and he gave it the name monkeypox (MONKEYPOX).
The first human case was described in 1970 in a nine‑month‑old infant in the Democratic Republic of the Congo, in the context of intensified smallpox surveillance programs (2).
On November 28, 2022, the World Health Organization (WHO) announced that it would begin to use the preferred term «mpox», instead of monkeypox as a synonym for monkeypox, due to social stigma and racist language, mainly in Africa.
ETIOLOGY:
The virus of MONKEYPOX belongs to the GENUS: ORTHPOXVIRUS; FAMILY: POXVIRIDAE, and two "clades" or variants have been described: the Central Africa clade or Clade I (formerly “Congo Basin”), associated with greater severity and lethality, and the West Africa clade or Clade II, of lower virulence (3).
DEFINITION OF CLADE, AND VARIANTS:
"A clade or variant is scientifically known as a group of organisms (or viruses) that descend from the same common ancestor and include all their known descendants, that is, they share the same characteristics; they are genetically distinct ‘families’ of the virus, with differences in lethality and mode of transmission."
- Variants or Clades of the Mpox Virus:
SOURCE: Spanish Society of Family and Community Medicine
The "clades" are mutations of the original virus, arising from changes in its genome, and the following have been described to date:
A.) CLADE I (Ia and Ib): Central Africa: More virulent; historical lethality up to ~10% in poor settings, especially children and unvaccinated individuals. Outbreaks more linked to contact with wild animals (zoonosis).
- Variant Clade Ia: "classical", Typical pattern: zoonosis from animals (rodents, wildlife) in the Democratic Republic of the Congo (DRC), and neighboring countries, with brief human outbreaks and limited person‑to‑person transmission, with a historical mortality in humans: range 5–10%, especially in children and unvaccinated individuals.
B.) CLADE II (IIa and IIb): West Africa: Less lethal (≈1–3% or less);
- Variant Clade IIa: local outbreaks, more zoonotic.
- Variant Clade IIb: This was the one that caused the global 2022 outbreak, because by mutating, it became better adapted to person‑to‑person transmission (especially via sexual contact), but its mortality is very low (<1%) in countries with good healthcare.
RESERVOIRS AND MODE OF TRANSMISSION:
Although it is true that THE MONKEYPOX VIRUS (mpox) was discovered in CRAB‑EATING MACAQUES (Cynomolgus Monkey), monkeys ARE NOT RESERVOIRS of the virus, but amplifiers, acting as accidental hosts (4).
The WHO has confirmed that the virus had circulated before 1958 in nature, and the main reservoirs (not 100% confirmed) correspond to AFRICAN RODENTS, including the Gambian rat (Cricetomys gambianus) and other small mammals among which stand out:
- Rope/string squirrels: (Funisciurus spp., especially F. anerythrus): Most probable reservoir according to ecological niche.
- Sun squirrels: (Heliosciurus spp.).
- African dormice: (Graphiurus spp.).
- Prairie dogs: (Chaetomys subspinosus).
OTHER POSSIBLES:
- Ground squirrels:
(Xerus spp.).
- Shrews: (Soricidae).
- Mice/rats:
generic African species.
Transmission to humans occurs through direct contact with skin lesions, bodily fluids, scabs, contaminated fomites, sexual intercourse, and also by exposure to short‑range aerosols.
In endemic settings, most cases derive from zoonosis, with a smaller proportion of person‑to‑person transmission (5).
In total, since MONKEYPOX in humans was first diagnosed in the Democratic Republic of the Congo in 1970, it has progressively appeared in other African countries and in countries outside the Asian continent.
This trend may be related to the end of vaccination against SMALLPOX CLASSIC SMALLPOX (smallpox), BECAUSE belonging to the same genus ORTHPOXVIRUS it conferred cross‑protection against MONKEYPOX (mpox). (5).
The incubation period of MONKEYPOX (mpox) in humans is 5 to 21 days, with a mean of 6–13 days, and the clinical picture is characterized by:
- A prodrome of fever, severe headache, malaise, myalgia, and lymphadenopathy (cervical, axillary, or inguinal).
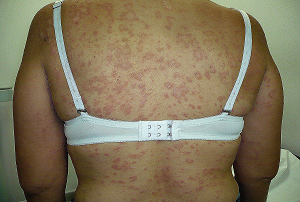
- Then a typical skin eruption appears, similar to CLASSIC SMALLPOX (6). The rash evolves from macules to papules, vesicles, and deep pustules, ultimately forming scabs over a course of 2–4 weeks.
- Symptoms such as odynophagia, cough, and, in severe cases, complications like pneumonia, sepsis, or encephalitis can occur (7).
SYMPTOMS IN MONKEYS:
In primates, especially macaques used in bio‑laboratories,
infection presents with fever, lethargy, generalized vesicular and
pustular skin lesions; in some experimental models, severe systemic
disease with high mortality may occur (8).
Under natural conditions, monkeys ARE NOT CONSIDERED THE PRIMARY RESERVOIR , as we explained previously, but amplifying hosts that can clinically show disease and are evident during outbreaks IN CAPTIVITY, or after intense exposure to the virus (9).
DIAGNOSTIC:
The diagnosis of MONKEYPOX (mpox), is made by:
A.) CLINICAL MANIFESTATIONS of the patient: Fever, general malaise, lymphadenopathy, and the rash, which begins with blisters, then pustules, which later turn into crusted‑like lesions or even ulcerations.
B.) PCR (Polymerase Chain Reaction) TEST: With which the viral DNA of mpox is detected, allowing differentiation of this virus from others such as VARICELLA‑ZOSTER virus (VZ).
The CDC recommends taking samples from 2 different lesions via swab, as well as from exudate from the pharynx, urethra, and anus (if no skin lesions are visible); these samples are processed in BSL‑2 laboratories, which can handle pathogens (bacteria, viruses) that may cause disease but, in general, are not easily transmitted by air in the social environment.
GEOGRAPHIC DISTRIBUTION:
Historically, since its discovery in 1958, MONKEYPOX has been endemic in countries of Central Africa (Clade I), West Africa (Clade II), with the Democratic Republic of the Congo (DRC) being the country that concentrates the majority of reported cases since 1970, when the first case was described in that country (10).
In 2022, ALARMS surged when the international spread of mpox was documented, with a multinational outbreak affecting Europe, North America, and other regions, driven by imported cases and subsequent community transmission (Clade IIb), caused by viral mutation with human‑to‑human transmission (7,9,10).
COUNTRIES AFFECTED 2022–2023
| Country (outside Africa) | Approx. cases 2022–2023 | Approx. % of world total | Approx. deaths 2022–2023 | Mortality (approx. CFR) |
|---|---|---|---|---|
| United States | ~31 000 | ~30–31 % | 40–50 | ~0.15 % |
| Spain | ~7 500 | ~7 % | 3–4 | ~0.05 % |
| Brazil | ~10 000 | ~9–10 % | 10–15 | ~0.1–0.15 % |
| France | ~4 000 | ~4 % | 1–2 | ~0.05 % |
| Germany | ~3 800 | ~3–4 % | 0–1 | ~0–0.03 % |
| United Kingdom | ~3 800–4 000 | ~4 % | 0–1 | ~0–0.03 % |
| Canada | ~1 500 | ~1–2 % | 0–1 | ~0–0.07 % |
| Peru | ~3 000 | ~3 % | 3–5 | ~0.1–0.15 % |
| Colombia | ~1 500 | ~1–2 % | 1–2 | ~0.07–0.13 % |
Note: For the first 5 months of 2022, approximately 1,405 cases and 62 deaths had already been reported in Africa in 4 countries (Cameroon, Central African Republic, Democratic Republic of the Congo, Nigeria), with a CFR (mortality) close to 4.4 %. This multinational outbreak belonged mainly to Clade IIb, lineage B.1
From 1970 to 2017–2021, Africa had already accumulated thousands of cases of mpox, especially in the Democratic Republic of the Congo (DRC), with hundreds of deaths; only between 2020 and 2021 12,457 cases and 365 deaths were recorded with a mortality of (CFR ≈2.9 %)
Now we will show you the evolution of MONKEYPOX (mpox) from the first human case detected in the Democratic Republic of the Congo (DRC) in 1970 up to today, 2026, so that you can see the progressive increase in cases.
HUMAN MPOX CASES IN AFRICA (1970–1986)
| Country | Period | Reported cases | % of total period | Reported deaths |
|---|---|---|---|---|
| Zaire / DRC | 1970–1979 | 38 (of 47) | ≈81 % | ≈7–8 (CFR ≈17 % global 1970–79) |
| Liberia, Sierra Leone, Nigeria, CAR* | 1970–1979 | ≈9 | ≈19 % | 0–1 (very limited data) |
| TOTAL 5 countries | 1970–1979 | 47 cases | 100 % | 8 deaths (CFR ≈17 %) |
| DRC (intensive WHO surveillance) | 1981–1986 | 338 (of 404 in Africa) | ≈84 % of total 1981–86 | Not well documented (underreporting) |
| Other Central and West African countries | 1981–1986 | ≈66 | ≈16 % | Not well documented |
| TOTAL AFRICA | 1981–1986 | 404 cases | 100 % | Deaths not specified (older WHO sources) |
HUMAN MPOX CASES IN AFRICA (1986–2021, SUMMARY)
| Country / Region | Period | Reported cases* | Approx. % of Africa | Reported deaths* |
|---|---|---|---|---|
| Democratic Republic of the Congo (DRC) | 1986–2000 | Sporadic cases; important underreporting (tens–hundreds) | Majority of known African cases | Few documented deaths; no reliable consolidated total |
| DRC | 2001–2017 | Thousands of suspected cases accumulated (several years with >2 000/year) | >70 % of African cases | Hundreds of deaths estimated (low–moderate CFR, underreporting) |
| Central African Republic (CAR) | 2001–2021 | 99 confirmed + 61 suspected cases (95 outbreaks, 40 confirmed) | Small fraction of total African cases | Very few deaths; no consolidated figure given |
| Nigeria | 2017–2018 | 122 confirmed/probable cases (276 suspected) | Important focus in West Africa | 7 deaths (CFR ≈6 %) |
| DRC | 2018–2021 | >3 000 suspected cases per year; peak of 6 216 cases and 222 deaths in 2020 | Clearly the majority of African cases in this period | At least 222 deaths in 2020; more deaths in other years |
| CAR | 2018–2021 | 79 confirmed cases in 7 localities (increase vs. previous years) | Small proportion, but increasing locally | Some deaths described; no precise global figure |
| Cameroon | 2018–2021 | 9 confirmed cases (no documented cases since 1989) | Very low proportion of total | No notable deaths reported in this period |
| Republic of the Congo (ROC) | 2018–2021 | 2 cases reported | Marginal | No deaths reported |
| Sierra Leone | 2018–2021 | 2 cases reported | Marginal | No deaths reported |
| AFRICA (countries with known cases) | 1986–2021 | Thousands of cases, concentrated in the DRC; notable outbreak in Nigeria (2017–2018) and increase in CAR since 2018 | DRC >70 % of total; Nigeria and CAR contribute in specific outbreaks/localities | Hundreds of deaths accumulated (at least 222 in the DRC in 2020 + 7 in Nigeria 2017–2018); the rest are underreported |
GLOBAL MPOX CASES REPORTED (2023–EARLY 2026, APPROX.)
| Year / Period | Approx. new cases | Approx. deaths (CFR) | Key comment |
|---|---|---|---|
| 2023 (January–November) | 10 000–15 000 |
Dozens of deaths within the 171 cumulative deaths by November
2023. Approx. global CFR: 0.2–0.3 %. |
Persistence of the global clade IIb outbreak with 500–1 000 cases per month. |
| Dec 2023 – 12 June 2024 (global) | ≈4 500 | Few additional deaths; global CFR ≈0.2–0.3 %. | Low but sustained circulation; clade IIb still dominates outside Africa. |
| 2024 (Africa, through 28 July) | 14 250 |
456 deaths (96 % in DRC). Regional CFR (Africa, period): ≈3 %. |
Sharp rise in DRC due to clade Ib; Africa becomes the main focus. |
| 12 June 2024 – 30 April 2025 (global) | ≈44 800 |
Hundreds of new deaths, mostly in Africa. Approx. global CFR: ~1 %. |
Prolonged wave linked to clade Ib, with high burden in rural African areas. |
| May 2025 – 31 January 2026 (global) | ≈37 400 |
Dozens–hundreds of additional deaths, concentrated in Africa. Approx. global CFR: ~0.5–1 %. |
Continued transmission in Africa; sporadic imported cases in other regions. |
| approximate total 2023 – 31 jan 2026 | 80 000–90 000 |
Hundreds of deaths (≥456 in Africa in 2024 + others in DRC and rest
of the world). Global CFR for the period: ~0.5 %. |
Decline after the 2022 peak, followed by a new African wave from clade Ib and sporadic outbreaks outside Africa. |
Now I will show you the UPDATED figures, up to FEBRUARY 2026, number of cases, percentage mortality, CLADES present, and comments.
MULTICOUNTRY MPOX OUTBREAK AND REGION OF THE AMERICAS – UNTIL FEBRUARY 2026
| Concept | Main figure | Details / comment |
|---|---|---|
| Global cases (2022–31 Jan 2026) | 179 612 confirmed cases | In 145 countries and territories. |
| Region of the Americas (2022–2026) | 73 641 confirmed cases | 160 deaths in 31 countries and territories; 41 % of the global total. |
| African region | 36.2 % of the global total | Contributes the second largest share of cases, with higher lethality. |
| European region | 17.8 % of the global total | Residual transmission, mainly in MSM networks, clade IIb predominant. |
| Region of the Americas – 2026 | 172 new cases |
Reported by 8 countries: Argentina, Bolivia (Plurinational State
of), Brazil, Canada, Chile, Colombia, Mexico and the United
States. To date, no deaths have been reported in 2026 in the region. |
| Clade Ib in the Americas (last 3 months) | 2 imported cases |
Mexico: n=1; Brazil: n=1. Two imported cases raise the region’s total accumulated clade Ib to 14. Both cases confirmed in travelers with prior exposure outside the country, who left before the test results were confirmed. |
| Countries with clade Ib (total accumulated) | 4 countries |
United States (n=9 cases), Canada (n=2 cases), Brazil (n=2 cases)
and Mexico (n=1 case). Total regional accumulated: 14 cases of clade Ib. |
| Summary | The Americas account for 41 % of the nearly 180 000 global cases accumulated. | Low mortality in 2026, with residual circulation of clade IIb and imported clade Ib cases, mainly detected in travelers. |
SOURCE: PAN AMERICAN HEALTH ORGANIZATION (PAHO): FEBRUARY 2026
In Venezuela there have been no reported cases since the first report in 1970, through February 2026, of Mpox. (Including the worldwide outbreak of 2022)
TREATMENT OF MONKEYPOX:
1. General management measures:
- Isolation and prevention of transmission:
The patient should be isolated until complete healing (detachment of scabs and formation of new skin), avoid physical contact, shared clothes and utensils, use a mask, and avoid sexual intercourse while active lesions are present (11).
- Symptom management: In general it is a self‑limiting disease, lasting about 2–4 weeks, and the following are used:
- Common analgesics (paracetamol, NSAIDs) for fever and pain, adequate oral hydration, rest, and skin hygiene: care of the skin to prevent secondary infections, control of itching or scratching, cleaning of wounds, and monitoring for complications, especially in cases of HIV and pregnancy (11).
2. VACCINES:
The vaccines used act both as prevention (to avoid infection) and as
post‑exposure prophylaxis (if given within the first two weeks after
contact with an active case). These are:
A.) JYNNEOS (Imvanex / Imvamune):
This vaccine was developed by the Danish pharmaceutical company
Bavarian Nordic before 2013, and is based on the modified vaccinia virus
Ankara‑Bavarian Nordic (MVA‑BN),
a live attenuated, non‑replicating virus designed to be safer than
traditional smallpox vaccines (12,13,).
It was approved for human use in 2013
in Canada and Europe (Imvamune/Imvanex) and in 2019 in the United States by the FDA (JYNNEOS), to prevent both smallpox and monkeypox (mpox)
(12).
- The vaccine is considered highly safe, even in immunocompromised individuals or those with dermatological disease. The standard regimen consists of 2 subcutaneous doses, with an interval of 28–30 days (4 weeks); to achieve full protection it is recommended to complete this schedule in people 18 years or older at high risk of exposure (men who have sex with men, healthcare workers, and contacts of cases).
- It induces cross‑protection against the virus of SMALLPOX (smallpox) and MONKEYPOX (mpox): reducing symptom severity, incidence, and transmission (12,13).
B.) ACAM2000:
- The ACAM2000 vaccine is a second‑generation vaccine against SMALLPOX (smallpox) developed by the company Acambis Inc., United Kingdom in the early 2000s; it was later acquired by the laboratory Sanofi Pasteur in 2008, and is currently manufactured for the USA by Emergent Product Development Gaithersburg, Inc., and approved for use in September 2007 by the FDA.
- This VACCINE was developedfrom the former Dryvax, the original vaccine against SMALLPOX used until the 1980s when smallpox was eradicated from the planet; its development was part of U.S. biodefense planning against bioterrorist attacks.
- In short, the ACAM2000 VACCINE is a "CLONE" of the Dryvax vaccine: a single viral particle was taken from it (which originally contained a mixture of calf‑lymph viruses, heterogeneous), it was amplified, and this resulted in a single cloned strain named ACAM1000, from which the ACAM2000 vaccine was created, genetically more uniform and stabilized.
- This vaccine is NOT USED for routine vaccination; it is reserved for cases of severe exposure (laboratory personnel), mpox or bioterrorist attacks, and a stockpile (strategic reserves) of about 100–200 million doses was created in the USA for bioterrorist attacks with the virus of SMALLPOX (smallpox). All of this was "created" after the 2001 attacks on the Twin Towers (14,15).
- Among the adverse effects or contraindications of this vaccine are: immunosuppression, atopic dermatitis, pregnancy, and others.
C.) ORAL TREATMENT: (antiviral drugs and oral antibiotics for outpatient use)
Because it is a self‑limiting disease, in most cases no specific treatment
is required; however, when there are patients at high risk—immunocompromised, children, pregnant women, extensive lesions—oral
drugs can be used, including:
- Tecovirimat (TPOXX): ST‑246
- An oral antiviral specific against ORTHOPOXVIRUSES, developed by SIGA Technologies, Inc., a U.S. pharmaceutical company focused on health and security (biodefense). The oral formulation was approved by the FDA in 2018, and the intravenous (IV) formulation in 2022 (16).
- It is not available over‑the‑counter (OTC), nor is it freely dispensed by prescription; access to this drug is coordinated through health departments and the CDC. It has been used in cases of MONKEYPOX (mpox), but it is also part of the strategic reserves against bioterrorism, created by the United States and Canada, specifically against attacks with the virus of SMALLPOX (smallpox).
- It is administered orally in divided doses over several days, and is specifically indicated for moderate to severe cases, or those at high risk of complications. It has shown to be safe and well tolerated in people with mpox. (16).
- Brincidofovir:
- This drug brincidofovir (CMX001, trade name TEMBEXA) was developed by the U.S. biopharmaceutical company Chimerix, Inc., based in Durham, North Carolina (17,18).
- It is an oral broad‑spectrum antiviral whose clinical development began in the 2000s, initially for infections with cytomegalovirus, adenovirus, and other DNA viruses in transplant recipients, and later extended as a possible countermeasure against ORTHOPOXVIRUSES (smallpox), in collaboration with the U.S. biodefense agency.
- The drug was approved on June 4, 2021 (during the COVID‑19 pandemic), under the name TEMBEXA(brincidofovir) in tablets and oral suspension for the treatment of SMALLPOX (smallpox), in adults and children (17,18).
- The approval was granted under the ANIMAL RULE (efficacy demonstrated in animal models of poxviruses), precisely because there are no natural smallpox cases in humans for clinical trials.
- In 2022–2023, Chimerix sold the worldwide rights to TEMBEXA (brincidofovir) for ORTHOPOXVIRUSES to Emergent BioSolutions, which keeps it as strategic reserve against bioterrorist attacks with the smallpox virus.
- It has limited utility in MONKEYPOX (mpox) due to its hepatic toxicity. In addition, its formal and specific indication is for SMALLPOX in humans (smallpox), which means that for mpox the preferred oral drug is TECOVIRIMAT. (16,17,18)
- Oral antibiotics for secondary infection:
- In cases with secondary bacterial infection of the lesions
(intense pain, redness, pus, fever), oral systemic antibiotics such as
cephalexin, ciprofloxacin, cefadroxil, or cloxacilin are used, according
to outpatient‑management guidelines issued by WHO, PAHO, and CDC.
CONCLUSIONS:
Despite the worldwide outbreak of 2022, todayMONKEYPOX (mpox), is under control, and the likelihood of a global pandemic is low, because it is a self‑limiting disease and because of the existing vaccines and available oral drugs.
- The estimated mortality for the Americas is 0.19%, for Europe 0.33%, and 5–10% in Africa; these percentages vary by region and "CLADE", with the zoonotic (Clade I) being more lethal than the human‑to‑human transmission (Clade IIb).
- However, the authorities from WHO, PAHO, and CDC maintain constant surveillance of this virus, which could eventually mutate into a more lethal generation or "clade".
Greetings to all,
Dr. José Lapenta
Dr. José M. Lapenta.
===============================================================
REFERENCIAS BIBLIOGRÁFICAS / BIBLIOGRAPHICAL REFERENCES
================================================================
11.) Guía Clínica para Manejo Ambulatorio de Viruela del Mono - MPX (PDF)
12.) Vaccines against monkeypox (2023)
13.) Effect of JYNNEOS vaccination on mpox clinical progression: a case-control study (2025).
14.) ACAM2000 Smallpox Vaccine (2024).
16.) Effectiveness of Tecovirimat in Mpox Cases: A Systematic Review of Current Evidence (2024).
17.) Brincidofovir in the Era of Mpox (2025).
18.) Brincidofovir: A Novel Agent for the Treatment of Smallpox (2023).
===================================================================
===================================================================
DATA-MÉDICOS/DERMAGIC-EXPRESS No 21-(250) 11/03/2.026 DR. JOSÉ LAPENTA
===================================================================
Produced by Dr. José Lapenta R. Dermatologist
Venezuela 1.998-2.026
Producido por Dr. José Lapenta R. Dermatólogo Venezuela 1.998-2.026
Tlf: 0414-2976087 - 04127766810
04243431100




















.png)